Medical Air USP
Medical Air USP is used extensively in the ICU, PICU and NICU areas, and with pulmonary nebulizers to reduce the risk of excess oxygen in the lungs or other body tissue (hyperoxia) during mechanical ventilation or surgical procedures.
| Purity Specifications | MSDS | |
|---|---|---|
| Purity: Oxygen 19.5 - 23.5% | Water and Oil: None | P-4560 |
| Balance Nitrogen | SO2≤5ppm | |
| Odor: None | NO+NO2≤2.5ppm | |
| CO≤10ppm | ||
| CO2≤500ppm |
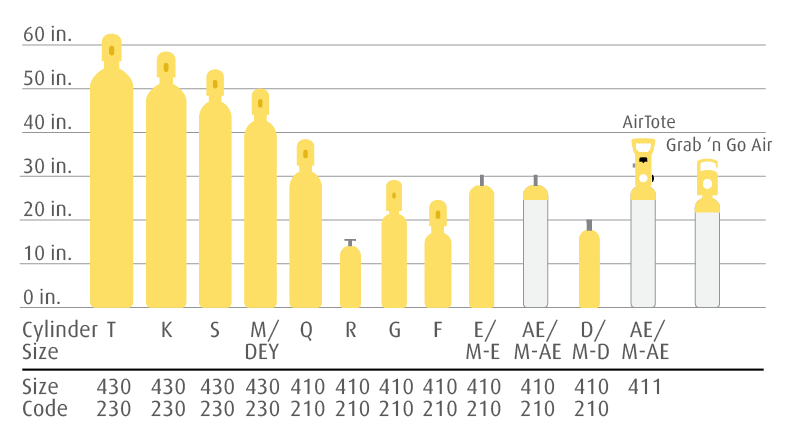
Cylinder Specifications & Equipment Recommendations
| Ordering Number | Cylinder Size | Nominal Contents | Pressure (psig) @ 70°F | CGA Connection | Regulator | |
|---|---|---|---|---|---|---|
| AI M-D | M-D | 14 cu ft | 2000 | 950 | WESM1-950-PG | |
| AI M-E | M-E | 23 cu ft | 2000 | 950 | WESM1-950-PG | |
| AI M-AE | AE | 22 cu ft | 2000 | 950 | WESM1-950-PG | |
| AI M-K | K | 232 cu ft | 2200 | 346 | WESM1-346-PG | WESM1-346-P |
| AI M-T | T | 310 cu ft | 2640 | 346 | WESM1-346-PG | WESM1-346-P |
| AI M-AEAIRTOTE | AE | 22 cu ft | 2000 | NR | AirTote, Aluminum | (Regulator Included) |




























